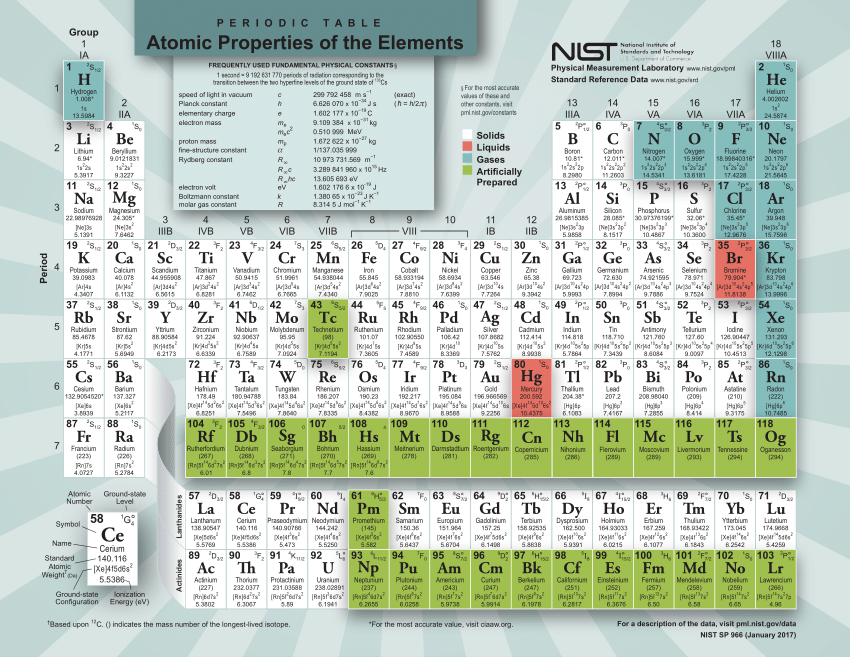 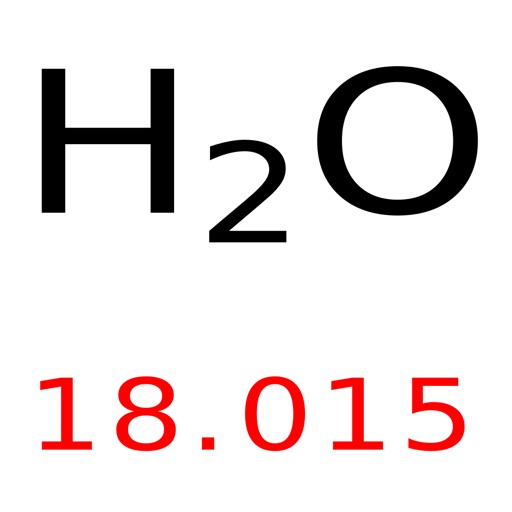 
The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. As hydrogen gas is formed of two hydrogen (H) molecules. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Trending Questions How are alluvial fans deltas natural levees and moraines formed? On earth the sun appears in the north or west or south or east.? What environmental issues does The Lorax face? What city rains the most in US? Where would you find particles of clay with no Sand or pebbles? How many kilos weigh a cubic yard of soil? What is the difference between relative dating and numerical? How does Canada's location climate and natural resources affected where people live? What is a way to control soil erosion in which farmers plant different crops? What tells how much rain has fallen? How many days until spring starts? What is the melting point of 24 gauge galvanized steel? What is a buckling zone? What does coastal vegetation mean? What term best describes rocky out-layer of Earth? What is a natural reservoir? Is the halite soft or hard? What gas stations in Little Rock Arkansas sell gasoline with no ethanol? What word means the same as earth? How many joules of heat are needed to raise the temperature of a 4.0 gram sample of water to 8. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, Mu 1 × 103 kgmol1. It can be calculated by multiplying the atomic weight of hydrogen (1.00784 atomic mass units) by 2. Multiplying the molar mass of each atom by the number of atoms of that type in bilirubins formula and adding the results.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
